Polytetrafluoroethylene (PTFE): Properties, Processing, and Applications
Polytetrafluoroethylene (PTFE), commercially known as Teflon, is a semi-crystalline fluoropolymer. PTFE is well-known for its application as a non-stick coating for kitchen pots and pans due to its exceptional heat and corrosion resistance. Besides this, these properties have proven beneficial in other industries too, namely the automotive, chemical and medical industries, where chemical inertness, electrical insulation and long service life are especially valuable.
PTFE has been the dominant fluoropolymer with a significant amount of market growth in recent years and is expected to gain yet more popularity in the coming years. The global revenue for PTFE is expected to reach more than US$7bn by 2022, and the demand is estimated to be as much as 247,000 megatonnes in the year 2022 [1]. This is due to increasing electronic and chemical applications, as well as the emerging application for polymers in photovoltaic modules and fuel cells.
Here you will learn about:
- Properties of PTFE
- Production and Processing of PTFE
- PTFE composites
- Applications of PTFE
- Example Grades of PTFE

Properties of PTFE
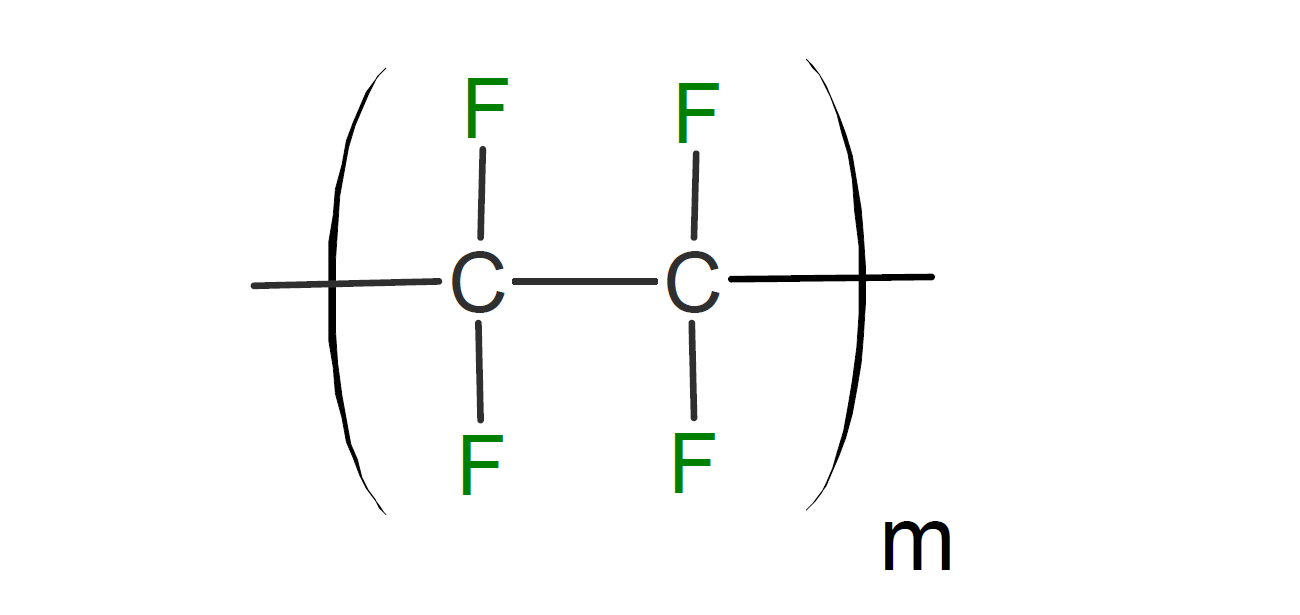
Figure 1. The structure of PTFE [2].
PTFE is a synthetic, semi-crystalline polymer made up of a carbon backbone surrounded by fluorine atoms. It is produced in a free-radical addition polymerisation reaction between tetrafluoroethylene (TFE) monomers. This layer of fluorine atoms acts as a barrier and is responsible for the high corrosion resistance and electrical inertness of PTFE. Additionally, the highly symmetrical structure of PTFE explains its hydrophobic nature, low surface tension and low dielectric constant. It is available as granules, fine powder or water-based dispersions depending on the application at hand.
Table 1. Properties of PTFE
|
Properties |
PTFE at 20 °C |
|
Physical Properties |
|
|
Density |
2.13 - 2.23 g/cm³ |
|
Mechanical Properties |
|
|
0.4 - 0.75 GPa |
|
|
350 - 550 % |
|
|
20 - 35 MPa |
|
|
Thermal Properties |
|
|
Melting Point |
325 - 335 ℃ |
|
1 - 2 E-4/K |
|
|
Maximum Service Temperature |
260 ℃ |
|
0.25 W/(m∙K) |
|
|
1000 J/(kg∙K) |
|
|
Flammability |
V0 UL 94 (Flame Retardant) |
|
Electrical Properties |
|
|
2.1 |
|
|
20 - 48 kV/mm |
|
|
1-1000 E+15 Ω∙m |
|
To explore the properties of PTFE in more detail and to compare it with other materials, visit the properties comparison page.
Production and processing of PTFE
PTFE can be found in granular, dispersion and fine powder forms. Semi-crystalline PTFE has high melting temperature and melt viscosity, making typical extrusion and injection moulding difficult. PTFE processing is, therefore, more similar to powder processing than that of traditional plastics.
Granular PTFE is produced in a water-based suspension polymerisation reaction. The resulting granular resin is often processed into shape through compression moulding. PTFE dispersion products are produced in a similar manner, with added dispersing agents. Dispersion products can be used for PTFE coatings or they can be processed into a thin film by film casting. PTFE powder is produced in an emulsion polymerisation reaction. The resulting fine powder can be paste extruded into PTFE tapes, PTFE tubing, and wire insulation, or used as an additive to increase corrosion resistance in other polymeric materials [3].
Fillers and additives for PTFE composites
Glass fibres can be added to PTFE between 5-40% to improve creep properties and general wear over time. Bronze is a common additive to improve thermal and electrical conductivity, increasing continuous service temperature. Carbon can be added to increase hardness and wear resistance. These additives are applicable to O-rings and seals that must be resistant to friction and durable in corrosive environments, used especially in automotive applications.
Applications of PTFE
Given that the specific processing of PTFE, including additive manufacturing, can have such a large impact on its properties, there is a large range of applications for this material. PTFE has applications in a huge range of industrial sectors, including automotive, chemical, electronic, aerospace and medical [2].
PTFE Sheet and Film
Given PTFE has such a high melting point for a polymer, it is commonly used to produce sheets and films for further applications. Sheets and thin films of PTFE are used mainly in the packaging sector. In the packaging of chemical equipment, pharmaceuticals and cosmetic products, a chemically inert material such as PTFE is worth the extra cost in comparison to other commodity packaging materials.
Industrial Coating
The most frequent application of PTFE globally is in industrial coatings. It has a very low friction coefficient and high-temperature resistance, making it perfect for demanding conditions. Interestingly, PTFE lubricant is used in the construction of igniters for rocket propellants, and to coat certain types of bullets to reduce wear on the inside of the barrel. Other uses include coatings for hoses subjected to corrosive environments and low-friction coatings on windscreen wipers and hair straighteners. Teflon coatings and non-stick wear are used on kitchenware to prevent food from sticking to pots and pans.
PTFE Powder
Fine PTFE powder can be paste extruded into useful shapes such as tapes, wires and tubes. These are mostly used for the insulation of wires and cables, tubing, pipe liners and films. PTFE powder can also be used as a powder coating for some materials, offering a more aesthetic finish.
Examples of Grades/Standards of PTFE
PTFE ISO-TECH
ISO-PTFE, also known as Teflon, offers a very high chemical and thermal resistance. In combination with its extremely low surface tension, ISO-PTFE is a very suitable material for a variety of challenging applications in the chemical, pharmaceutical and food industries.
PPS GF40 PTFE SG401E65
This is a 40% glass fibre reinforced PPS & PTFE compound, specially formulated to have a low coefficient of friction for use in low surface friction and wear applications.
PEEK PTFE SG201RN
This is a PTFE & PEEK alloy grade specially formulated to have a low coefficient of friction for use in low surface friction and chemical resistance.
PEEK CF30 G PTFE SGC301E4
This is a 30% carbon-fibre reinforced PTFE and graphite-reinforced PEEK compound developed to provide excellent wear resistance, lower coefficient of friction and better chemical resistance.

Sources
[1] "PTFE Properties, Production, Price, Market and Uses", Plastics Insight. [Online].
[2] "POLYTETRAFLUOROETHYLENE (PTFE)" (2018), Polymer Properties Database. [Online].
[3] “Manufacturing Polytetrafluoroethylene” (2013), in Introduction to Fluoropolymers: Materials, Technology and Applications, Ed. S. Ebnesajjad, William Andrew, pp.91–124. [Online].